Many people assume Anhydrous Hydrofluoric Acid is made by simply “concentrating hydrofluoric acid” or by dissolving fluorine into another chemical system, but that is not how industrial AHF production works. That misunderstanding leads to poor technical writing, weak procurement judgment, and dangerous oversimplification of one of the most important fluorine-industry raw materials. In reality, AHF is produced through a specialized inorganic chemical process that starts with acid-grade fluorspar and sulfuric acid, then moves through reaction, gas cleanup, condensation, scrubbing, and final distillation to achieve a very high-purity anhydrous product. Because hydrogen fluoride is highly corrosive, volatile, and toxic, every part of the manufacturing chain is designed around containment, impurity control, and recovery efficiency rather than simple “acid production” logic. (US EPA)
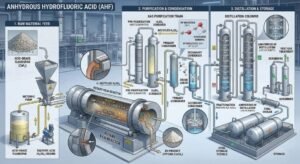
Anhydrous Hydrofluoric Acid (AHF / HF) is made industrially by reacting acid-grade fluorspar, which is mainly calcium fluoride (CaF2), with sulfuric acid (H2SO4). The core reaction is CaF2 + H2SO4 → CaSO4 + 2HF. In a typical plant, dry fluorspar and a slight excess of sulfuric acid are fed into a heated reactor system, commonly a prereactor and horizontal rotary kiln operated around 200 to 250°C. The reaction releases hydrogen fluoride-containing gas, which is cleaned of particulates, partially condensed, scrubbed, and then distilled. The final distillation step produces hydrofluoric acid at about 99.98% purity, which is sold as anhydrous hydrogen fluoride, while weaker commercial hydrofluoric acid grades are prepared later by controlled dilution with water. (US EPA)
If you want to understand AHF manufacturing correctly, the key is to look at the full process chain rather than just the reaction equation. The chemistry itself is simple on paper, but commercial production depends on ore quality, feed drying, reaction temperature, gas handling, condenser performance, impurity removal, by-product management, distillation design, and strict materials-of-construction discipline. That is why two suppliers can both claim to produce AHF, while only one delivers the purity, stability, and packaging performance that serious fluorochemical or industrial users actually need. (US EPA)
Anhydrous hydrofluoric acid is made by simply removing water from ordinary aqueous hydrofluoric acid.False
Commercial AHF is produced primarily by reacting acid-grade fluorspar with sulfuric acid, then purifying the resulting hydrogen fluoride through condensation, scrubbing, and distillation. It is not generally made by merely drying standard aqueous hydrofluoric acid.
The industrial starting point: fluorspar, not dilute hydrofluoric acid
The most important fact about AHF production is that the process starts with fluorspar, also called fluorite, rather than with a pre-existing bottle or tank of hydrofluoric acid. ATSDR identifies fluorite, or calcium fluoride, as the most important natural starting material for fluorine chemicals, including hydrogen fluoride. EPA’s hydrofluoric acid process section states directly that hydrofluoric acid is manufactured by the reaction of acid-grade fluorspar (CaF2) with sulfuric acid. In other words, industrial AHF production begins with a mineral feedstock, not a recycled liquid acid stream. (US EPA)
That distinction matters for buyers and marketers because it explains where AHF sits in the value chain. It is not a casual derivative chemical produced from generic upstream acids; it is one of the primary conversion products that turns mineral fluorine into industrially useful reactive fluorine. This is why the economics of AHF are tied closely to the availability and quality of acid-grade fluorspar, as well as to sulfuric acid supply, plant purification capability, and downstream fluorochemical demand. When the fluorspar market changes, the AHF market usually feels it. (US EPA)
Acid-grade fluorspar is important because the starting ore quality directly affects plant efficiency and product quality. Lower-grade feed can raise impurity loads, worsen solids handling, increase downstream cleanup burden, and reduce overall process stability. While the simplified reaction equation only shows CaF2 and H2SO4, real industrial ore contains impurities that influence dust generation, silicon-containing side reactions, residue composition, and scrubber load. That is one reason AHF manufacturing is a process-engineering discipline, not just a stoichiometric chemistry exercise. (US EPA)
Main raw materials used to make AHF
| Raw material | Role in the process | Why it matters |
|---|---|---|
| Acid-grade fluorspar (CaF2) | Primary fluorine source | Determines HF yield potential and impurity burden |
| Sulfuric acid (H2SO4) | Reactant that liberates HF from CaF2 | Drives the core conversion reaction |
| Heat / external firing | Supplies energy for the endothermic reaction | Maintains reaction temperature and throughput |
| Cooling / refrigeration | Condenses HF from process gas | Enables crude HF recovery |
| Water / caustic in scrubbers | Captures residual HF and by-products | Reduces emissions and recovers additional fluorine value |
The raw-material picture also explains why AHF is often described as a platform fluorine intermediate. Fluorine is mined in mineral form, converted to hydrogen fluoride, and then distributed into downstream fluorochemicals, fluoride salts, catalytic systems, and specialty materials. From an industrial perspective, AHF is one of the main bridges between ore and advanced chemistry. (US EPA)
The core reaction that makes AHF
The main industrial reaction is straightforward:
CaF2 + H2SO4 → CaSO4 + 2HF
EPA presents this exact equation in its hydrofluoric acid process documentation. On paper, it looks simple: calcium fluoride reacts with sulfuric acid to produce calcium sulfate and hydrogen fluoride. But the commercial reality is more complicated because the process must deal with solids handling, heat transfer, gas evolution, entrained dust, side reactions, condensation, and purification. So while the reaction equation is foundational, it is only the beginning of the story. (US EPA)
The reaction is described by EPA as endothermic, which means it requires heat input rather than generating enough heat on its own. EPA says the reaction typically takes 30 to 60 minutes in horizontal rotary kilns externally heated to about 200 to 250°C. This is a critical detail because it shows that industrial AHF production is a high-temperature solid-acid reaction, not a room-temperature mixing step. The plant must continuously deliver raw materials, maintain residence time, and keep the reactor hot enough to drive conversion and gas release efficiently. (US EPA)
Because HF is produced as part of a hot gas mixture, the reaction zone is only one part of the process. The downstream sections must immediately take over to separate entrained solids, remove sulfuric acid mist and water, condense hydrogen fluoride, recover residual fluorine values, and purify the product. That is why experienced engineers do not talk about “the reactor” as the whole plant. They talk about the reaction-and-recovery train. The valuable product is not simply created in the kiln; it is created across the kiln, the gas cleanup train, the condensation system, and the final distillation section together. (US EPA)
Core reaction summary
| Item | Industrial meaning |
|---|---|
| CaF2 | Mineral fluorine feedstock from acid-grade fluorspar |
| H2SO4 | Acid reactant that displaces HF from calcium fluoride |
| CaSO4 | Solid by-product removed from the reactor discharge |
| HF | Desired gaseous product, then condensed and purified |
| Endothermic reaction | External heat is required to maintain production |
This is also why AHF plants are not trivial to scale. You need solids feeding, acid dosing, rotary or equivalent reaction technology, gas-solid separation, condensation, scrubbing, distillation, and corrosion-resistant containment all working together. (US EPA)
What happens in the reactor section
According to EPA, a typical plant uses a stationary prereactor and/or a horizontal rotary kiln. Dry fluorspar and a slight excess of sulfuric acid are fed continuously to the front end of the system, often by screw conveyor. The prereactor’s job is to mix the feed components before charging to the kiln, and the kiln provides the heated residence time needed for the reaction to proceed. Calcium sulfate leaves the opposite end through an air lock, while hydrogen fluoride-containing gas exits from the front end. (US EPA)
That reactor arrangement reveals several practical truths about AHF production. First, the plant is built around continuous operation, not only batch chemistry. Second, dry feed handling is essential, because excess moisture would disrupt control and increase the burden on downstream purification. Third, the process is fundamentally a solid–liquid–gas conversion system: mineral solids and liquid sulfuric acid react under heat to release gaseous hydrogen fluoride. That phase change is one reason AHF manufacturing looks more like a thermal process plant than a simple liquid-acid blending facility. (US EPA)
The kiln section also produces more than just HF and calcium sulfate. EPA notes that the gas stream can contain excess sulfuric acid from the primary reaction, plus silicon tetrafluoride (SiF4), sulfur dioxide (SO2), carbon dioxide (CO2), water, and entrained particulate generated through secondary reactions and feed impurities. This is extremely important because it explains why purification is not optional. The reactor does not directly produce finished anhydrous HF; it produces a dirty, mixed gas stream that must be engineered into a purified chemical product. (US EPA)
From a plant-design viewpoint, the reactor section therefore has four simultaneous jobs:
- convert CaF2 into HF efficiently
- manage temperature and residence time
- discharge solid by-product safely
- send a recoverable gas mixture to downstream cleanup
If any one of those jobs is unstable, the final AHF quality and yield will suffer. (US EPA)
Why the gas cleanup train is essential
After the reactor, the process enters what may be the most underestimated part of AHF manufacturing: gas treatment and product recovery. EPA states that entrained particulates are first removed by a dust separator and returned to the kiln. Sulfuric acid and water are then removed in a precondenser, after which hydrogen fluoride vapors are condensed in refrigerant condensers to form crude HF. The remaining gas stream goes through a sulfuric acid absorption tower or acid scrubber to remove most of the remaining HF and residual sulfuric acid, then through water scrubbers where SiF4 and remaining HF are recovered as fluosilicic acid (H2SiF6). Tail gases finally pass through a caustic scrubber before release. (US EPA)
This sequence shows that commercial AHF production is, in large part, a separation and recovery process. The reaction may create the hydrogen fluoride, but the cleanup train determines how much of that value is recovered, what impurities remain, what by-products are captured, and how cleanly the plant operates. In other words, two plants with similar reactors can produce very different commercial outcomes depending on their gas cleanup and purification capability. (US EPA)
The cleanup train also creates economic value beyond AHF itself. EPA notes that the water scrubbers recover silicon tetrafluoride and remaining HF as fluosilicic acid, which is a commercially relevant fluoride-containing by-product in some industrial contexts. So the plant is not simply removing contaminants; it is recovering fluorine value from side streams that would otherwise be wasted or become more expensive emissions liabilities. Strong plants usually think in terms of fluorine recovery efficiency, not just headline HF output. (US EPA)
Simplified process flow of AHF manufacturing
| Process section | Main task | Output |
|---|---|---|
| Raw material feed | Deliver dry fluorspar and sulfuric acid | Controlled input to reactor |
| Prereactor / rotary kiln | Generate hydrogen fluoride from CaF2 + H2SO4 | Hot gas + CaSO4 solids |
| Dust separation | Remove entrained particulate | Cleaner gas, solids returned |
| Precondenser | Remove sulfuric acid and water | Cleaner HF-rich gas |
| Refrigerant condenser | Condense hydrogen fluoride | Crude HF |
| Acid scrubber | Recover remaining HF and acid traces | Additional HF recovery |
| Water scrubber | Capture SiF4 and residual HF | Fluosilicic acid stream |
| Caustic scrubber | Polish tail gas before release | Reduced emissions |
| Distillation | Upgrade crude HF to anhydrous grade | AHF around 99.98% purity |
This is the part many basic product pages leave out, but for serious readers it is one of the most useful sections. It explains why AHF plants are not only chemical plants, but also sophisticated gas-handling and purification systems. (US EPA)
How crude HF becomes anhydrous HF
The product leaving the condenser is not yet the final commercial AHF grade. EPA states that hydrogen fluoride and sulfuric acid are delivered from intermediate storage tanks to distillation columns, where hydrofluoric acid is extracted at 99.98 percent purity. This is the decisive finishing step. Distillation is what converts crude, recovery-grade HF into high-purity anhydrous hydrogen fluoride suitable for demanding industrial uses. (US EPA)
This point is central to understanding AHF manufacturing: AHF is not just “generated,” it is purified into specification. The final product must meet tight expectations for water content and overall purity. That is why the distillation system is commercially as important as the kiln. A plant that reacts fluorspar and sulfuric acid successfully but lacks strong distillation control may still fail to produce premium AHF. The market pays for usable purity, not just gross HF generation. (US EPA)
EPA also notes that weaker concentrations, typically 70 to 80 percent, are prepared by dilution with water after the high-purity product is obtained. That fact is helpful because it corrects a common misconception. Lower-strength commercial hydrofluoric acid products are often made from the purified HF stream; the plant does not normally begin with low-strength HF and dry it backwards into AHF. The sequence is the opposite: produce high-purity HF first, then dilute deliberately when lower strengths are needed. (US EPA)
For buyers, this matters because it explains why anhydrous HF is the upstream master grade in many production systems. Once you can reliably make purified AHF, you can generate other HF concentrations as downstream products. The reverse is not the usual industrial foundation. (US EPA)
What “anhydrous” really means in manufacturing terms
In commercial language, “anhydrous” means the product contains very little water and behaves as a highly concentrated hydrogen fluoride product rather than an aqueous acid solution. Honeywell’s SDS for hydrogen fluoride (100%) lists the substance as a colourless fuming liquid with a boiling point of 19.5°C and a melting point around -83°C, which reflects the physical behavior of the purified anhydrous product. Those physical properties help explain why dryness control and sealed containment are so important after distillation. (prod-edam.honeywell.com)
In manufacturing terms, “anhydrous” means several things at once:
- the product must be kept essentially water-free after purification
- equipment and transfer systems must minimize moisture ingress
- packaging must preserve grade integrity during storage and shipment
- downstream users can treat the product as a dry HF source rather than a water-rich acid solution
This is why AHF manufacturing does not end at the distillation column. It extends into storage, loading, transport packaging, and documentation. A technically good distillation result can still become a commercially bad shipment if moisture contamination, packaging incompatibility, or transfer losses degrade the product before it reaches the customer. (US EPA)
The role of by-products and side streams
AHF plants do not operate in a perfectly clean single-reaction universe. EPA’s process description shows that side streams and by-products are an inherent part of the process. The most obvious solid by-product is calcium sulfate (CaSO4), which leaves the kiln. The gas treatment train also captures fluorine-bearing side streams, especially fluosilicic acid, formed when SiF4 and remaining HF are scrubbed in water. (US EPA)
This matters for both process engineering and business logic. From the engineering side, by-products must be controlled so they do not foul the process, overload scrubbers, or distort purification. From the business side, by-products can affect plant economics. A facility that recovers fluorine efficiently and finds value in side streams can outperform a plant that treats everything beyond crude HF as waste. In industrial minerals and inorganic chemical manufacturing, these differences can be material to profitability. (US EPA)
A useful way to think about an AHF plant is that it is not just making one molecule. It is splitting a mineral resource into multiple process streams:
- premium purified HF
- diluted HF products, if desired
- calcium sulfate solid residue
- fluorosilicic-acid-containing recovery streams
- scrubbed off-gases that must meet emission controls
That systems view is more realistic than imagining a single pipe delivering finished AHF from one reactor. (US EPA)
Why purification quality determines supplier quality
From a commercial standpoint, the most important difference between an ordinary AHF producer and a strong one is often purification discipline rather than reaction chemistry alone. The core chemistry is well known. What separates suppliers is how consistently they can manage feed variability, reaction conditions, crude HF recovery, distillation purity, packaging quality, and shipment stability. EPA’s process summary shows that final hydrofluoric acid purity of about 99.98% depends on a dedicated distillation stage after multiple upstream cleanup steps. That implies that product quality is the result of an integrated manufacturing system, not a single reaction result. (US EPA)
For customers in fluorochemicals, high-performance inorganic fluoride production, or other moisture-sensitive applications, this is critical. Small differences in water content, residue, or impurity carryover can affect yield, reaction selectivity, catalyst behavior, equipment corrosion, and product qualification. So when a supplier says “we make AHF,” the real question is not only whether they run the reaction. It is whether they can deliver repeatable spec-grade AHF lot after lot. (US EPA)
What separates strong AHF production from weak AHF production
| Production factor | Weak plant outcome | Strong plant outcome |
|---|---|---|
| Raw material control | Variable ore quality, unstable operation | Consistent feed preparation and better conversion |
| Reactor stability | Poor residence control, more side burden | Predictable gas generation and solids discharge |
| Gas cleanup | Higher impurity carryover | Better crude HF quality |
| Distillation | Inconsistent purity | Stable high-purity AHF |
| Recovery efficiency | More fluorine loss to waste | Higher yield and better economics |
| Packaging discipline | Moisture pickup and shipment risk | Better preserved anhydrous quality |
| Environmental control | Higher scrubber/emission stress | Cleaner, more controlled operation |
This is also why technical buyers should look beyond a one-line COA. The best suppliers usually understand the full manufacturing chain and can discuss feed purity, product grade, loading practices, compatibility, and application fit in detail. (US EPA)
Environmental control is built into the process, not added at the end
Because HF is highly hazardous, emissions control is not a side issue. EPA’s hydrofluoric acid process description makes clear that condensing, scrubbing, and absorption equipment suppress emissions to a great extent and are part of normal product recovery and purification. In other words, environmental control and product recovery are closely linked in AHF manufacturing. The same equipment that helps clean the gas stream also helps recover valuable HF and fluorine-containing by-products. (US EPA)
This is an important design truth. In many chemical plants, environmental control is partly an end-of-pipe question. In AHF manufacturing, it is much more deeply integrated into the core process. If you do not control the gas streams well, you do not just risk compliance problems; you also lose product, reduce yield, degrade process efficiency, and put the whole plant at greater hazard. So emissions control in AHF production is not merely a legal obligation. It is a basic production discipline. (US EPA)
For readers building industrial content, this section is worth highlighting because it makes the manufacturing story more realistic. Serious AHF production is always tied to condensers, scrubbers, recovery sections, and tight containment. Any explanation that skips those units and jumps straight from “fluorspar + sulfuric acid” to “finished AHF” is incomplete. (US EPA)
The practical sequence of making AHF
To make the production logic easier to follow, here is the process in a clean operational sequence.
Step 1: Prepare the mineral and acid feed
The plant starts with dry acid-grade fluorspar and sulfuric acid. The raw materials are metered and introduced into a prereactor and/or rotary kiln system. EPA notes that dry spar and a slight excess of sulfuric acid are continuously fed, typically using a screw conveyor arrangement. The dryness of the feed matters because uncontrolled moisture would increase downstream purification burden and can complicate gas handling. (US EPA)
Step 2: Drive the CaF2–H2SO4 reaction under heat
The mixed feed reacts in a heated system, usually around 200 to 250°C, with a residence time of about 30 to 60 minutes. The reaction is endothermic, so external heating is required. Solid calcium sulfate leaves the kiln, while HF-containing gas exits toward the cleanup train. This is the fundamental conversion step that liberates hydrogen fluoride from the mineral fluorine source. (US EPA)
Step 3: Remove dust, sulfuric acid, and water from the gas stream
Before HF can be condensed effectively, the plant removes entrained particles and strips out sulfuric acid and water using dust separation and precondensation. This protects downstream condensation and purification equipment and improves crude HF quality. It also helps stabilize overall recovery performance. (US EPA)
Step 4: Condense and recover crude HF
The cleaned gas then passes through refrigerant condensers, where hydrogen fluoride is condensed into crude HF. Additional recovery happens in acid scrubbers, while water scrubbers capture residual fluorine values as fluosilicic acid. This step is where the hot gas stream is transformed into recoverable liquid HF and secondary fluoride-containing streams. (US EPA)
Step 5: Distill crude HF into high-purity AHF
The crude HF stream is sent to distillation columns, where purified hydrofluoric acid is recovered at about 99.98% purity, according to EPA. This is the formal point at which the product becomes commercial anhydrous hydrogen fluoride rather than crude process HF. Lower-strength commercial hydrofluoric acid products can then be made by controlled water dilution if needed. (US EPA)
Step 6: Store and ship under anhydrous conditions
Once purified, AHF must be held in compatible, sealed systems that preserve dryness and control vapor release. Honeywell’s SDS identifies the product as a fuming liquid with a boiling point of 19.5°C, which explains why storage and transfer design remain part of the manufacturing challenge even after purification is complete. (prod-edam.honeywell.com)
That stepwise view is useful because it separates the process into what really matters operationally: feed preparation, reaction, gas conditioning, product recovery, final purification, and protected storage. (US EPA)
What equipment is typically involved in AHF production
The plant equipment implied by EPA’s process description is broader than many readers expect. AHF manufacture typically involves:
- raw material storage and feed systems
- screw conveyors or controlled solids transfer systems
- prereactors
- externally heated horizontal rotary kilns
- air-lock discharge for solids
- dust separators
- precondensers
- refrigerant condensers
- sulfuric acid absorption towers or acid scrubbers
- water scrubbers
- caustic scrubbers
- intermediate storage tanks
- distillation columns
- final storage and transfer systems for purified HF
Each of these units exists for a reason. The kiln makes HF possible, but the rest of the plant makes AHF saleable, recoverable, and controllable. This is one reason why AHF production capacity is not easy to build casually. Even if the chemistry is old and well established, the equipment train remains specialized and demanding. (US EPA)
Equipment purpose chart
| Equipment | Main purpose in AHF production |
|---|---|
| Prereactor | Mix raw materials before kiln entry |
| Rotary kiln | Provide heated reaction zone |
| Dust separator | Remove entrained solids from gas |
| Precondenser | Strip sulfuric acid and water |
| Refrigerant condenser | Convert HF vapor into crude liquid HF |
| Acid scrubber | Recover remaining HF and acid traces |
| Water scrubber | Capture residual fluorine species as H2SiF6 |
| Caustic scrubber | Final gas cleanup before release |
| Distillation column | Produce high-purity anhydrous HF |
| Storage system | Preserve grade and control vapor risk |
Why AHF manufacturing is difficult even though the equation is simple
A recurring mistake in technical writing is to assume simple stoichiometry means simple production. AHF is the opposite. The chemistry equation is concise, but the plant is demanding because the process combines:
- mineral feed variability
- high-temperature reaction control
- corrosive and toxic gas handling
- volatile product recovery
- impurity and moisture management
- high-purity distillation
- by-product capture
- strict containment requirements
That combination is what makes AHF manufacturing industrially serious. It is not enough to know the formula HF. A producer must know how to turn ore into high-purity liquid hydrogen fluoride safely and consistently. EPA’s process flow and ATSDR’s production summary together make that clear: the raw chemical source is mineral fluorite, but the commercial product emerges only after engineered reaction and purification. (US EPA)
This is also why AHF plants are tightly connected to downstream fluorochemical ecosystems. Once a plant can make stable AHF, it can feed many other fluorine-value chains. Once it cannot, those downstream chains become vulnerable. So the manufacturing question is not just “How is AHF made?” It is also “How is the fluorine industry supplied?” In many cases, the answer begins with the same fluorspar-to-HF process described here. (atsdr.cdc.gov)
The relationship between AHF and weaker hydrofluoric acid grades
Readers often confuse AHF with hydrofluoric acid solutions such as 49%, 55%, or 70% HF. The manufacturing logic helps clear that up. EPA states that after distillation, hydrofluoric acid is extracted at 99.98 percent purity, and weaker concentrations are then prepared by dilution with water. That means the anhydrous or near-anhydrous product is the primary purified output, and lower-strength grades are derived from it. (US EPA)
This is a commercially useful point because it tells buyers how product families are structured. Anhydrous HF sits at the top of the purity ladder as the dry master product. Aqueous hydrofluoric acid grades are then produced when application needs call for a solution product. So if a user needs a wet etching acid, the supplier may dilute purified HF. If the user needs dry fluorination chemistry, the supplier preserves the product in anhydrous form. That is why the manufacturing route and the application route are closely connected. (US EPA)
Common misconceptions about how AHF is made
Several misconceptions show up repeatedly in industrial content and procurement conversations.
Misconception 1: AHF is just concentrated hydrofluoric acid
That is not the standard industrial picture. Commercial AHF is produced from fluorspar + sulfuric acid, then purified. Weaker HF grades are generally derived afterward by dilution, not the other way around. (US EPA)
Misconception 2: The reactor alone makes finished AHF
Also false. The reactor makes a mixed gas stream containing HF plus other substances. Product-grade AHF only emerges after dust removal, condensation, scrubbing, and distillation. (US EPA)
Misconception 3: AHF production is just one reaction vessel and one storage tank
EPA’s process description shows a much broader system involving prereactor, kiln, separators, multiple scrubbers, condensers, storage, and distillation. (US EPA)
Misconception 4: By-products are irrelevant
Not true. Calcium sulfate, fluorosilicic acid, and tail-gas streams all affect process economics, emissions control, and fluorine recovery. (US EPA)
The market reality is stricter. High-purity AHF requires strong purification, handling discipline, and dryness control, not only a reaction section. (US EPA)
What buyers should ask an AHF manufacturer
If you are sourcing AHF, the production method should shape your qualification questions. A serious supplier should be able to answer points such as:
- What raw material quality is used for the fluorspar feed?
- How is the reaction section configured?
- How is crude HF purified and what distillation capability is available?
- What is the guaranteed purity of the final AHF?
- How is moisture controlled during storage and loading?
- What by-product recovery and scrubbing systems are used?
- How stable is lot-to-lot quality?
- What packaging formats are available for preserving anhydrous grade?
These are not abstract questions. They go directly to the manufacturing chain described by EPA and to the physical properties described in the SDS. If a supplier cannot discuss how the product is actually made and preserved, that is usually a warning sign for technical buyers. (US EPA)
Final answer in plain language
Anhydrous Hydrofluoric Acid is made industrially from acid-grade fluorspar (calcium fluoride) and sulfuric acid. The two raw materials react in a heated process, typically involving a prereactor and externally heated rotary kiln, to produce hydrogen fluoride gas and calcium sulfate. The gas then passes through dust removal, precondensation, condensers, scrubbers, and finally distillation. That distillation step upgrades crude hydrogen fluoride into high-purity AHF, typically around 99.98% purity, while lower-strength hydrofluoric acid products are made later by adding water in a controlled way. (US EPA)
So the real answer to “How is AHF made?” is not just one equation. It is a full industrial chain: mineral feedstock selection, thermal reaction, gas cleanup, condensation, fluorine recovery, and high-purity distillation. That is what turns a fluorine-bearing ore into one of the world’s most important fluorochemical raw materials. (US EPA)
Let’s talk about the part that really matters
If your team is evaluating Anhydrous Hydrofluoric Acid, hydrofluoric acid grades, or upstream fluorochemical raw materials, the most important question is not only whether a supplier can quote HF, but whether they can explain how the product is made, purified, and preserved to fit your actual application. At Sparrow-Chemical, we focus on practical industrial communication around fluorochemicals and raw-material selection logic. For HF-related product discussions, specification matching, or fluorochemical sourcing inquiries, visit Sparrow-Chemical: https://sparrow-chemical.com/