Fluorochemicals play a critical role in modern industry, yet many people—even in technical fields—struggle to clearly define what they are. These compounds appear everywhere: semiconductor fabrication, lithium-ion batteries, aerospace coatings, pharmaceuticals, refrigerants, and high-performance plastics. Despite their widespread use, the term “fluorochemical” is often misunderstood or confused with specific subcategories such as PFAS, fluoropolymers, or refrigerants. This confusion can lead to poor purchasing decisions, regulatory misunderstandings, or incorrect technical assumptions about material performance. Understanding what fluorochemicals truly are—and why they possess such extraordinary properties—is essential for engineers, chemists, and industrial buyers.
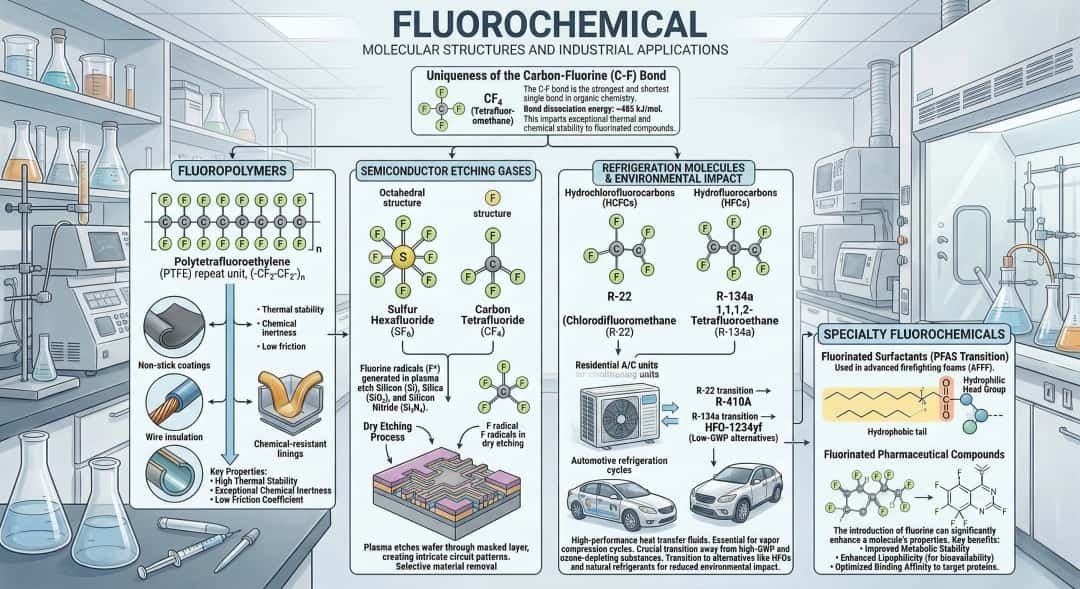
A fluorochemical is any chemical compound that contains at least one carbon-fluorine (C–F) bond, one of the strongest bonds in organic chemistry. This bond gives fluorochemicals unique properties such as exceptional chemical resistance, thermal stability, low surface energy, and high electrical insulation. Because of these characteristics, fluorochemicals are widely used in industries including electronics, pharmaceuticals, aerospace, energy storage, and refrigeration. The category includes fluoropolymers, fluorinated gases, fluorinated solvents, fluorinated intermediates, and fluorosurfactants.
To fully understand fluorochemicals, it is important to examine their chemical structure, major product categories, industrial applications, production methods, and the scientific reasons behind their unusual physical and chemical behavior. This deeper perspective explains why fluorochemicals have become indispensable materials across modern technology and advanced manufacturing.
The Chemical Definition of Fluorochemicals
At its most fundamental level, a fluorochemical is defined by the presence of fluorine atoms bonded to carbon atoms.
Key Structural Characteristics
Fluorochemicals differ from conventional organic molecules because fluorine is:
- the most electronegative element
- extremely small in atomic radius
- capable of forming highly stable bonds with carbon
When fluorine replaces hydrogen atoms in hydrocarbons, the resulting molecules exhibit dramatically different properties.
Comparison of Carbon Bonds
| Bond Type | Bond Energy (kJ/mol) | Chemical Stability |
|---|---|---|
| C–C | ~348 | Moderate |
| C–H | ~413 | Stable |
| C–Cl | ~338 | Moderate |
| C–F | ~485 | Extremely stable |
The strength of the carbon-fluorine bond is the main reason fluorochemicals exhibit exceptional durability.
Effects of Fluorine Substitution
Replacing hydrogen atoms with fluorine produces several important changes:
- increased thermal stability
- improved chemical resistance
- reduced surface energy
- higher dielectric strength
- greater hydrophobicity and oleophobicity
These properties are why fluorochemicals are widely used in extreme environments.
Major Categories of Fluorochemicals
Fluorochemicals include a wide variety of compounds used across different industries.
Classification of Fluorochemicals
| Category | Typical Examples | Key Applications |
|---|---|---|
| Fluoropolymers | PTFE, PVDF, FEP, PFA | Non-stick coatings, electronics |
| Refrigerants | HFCs, HFOs | Air conditioning and cooling |
| Fluorosurfactants | PFOS alternatives | Firefighting foams, coatings |
| Fluorinated solvents | HFE fluids | Electronics cleaning |
| Fluorinated intermediates | Fluorobenzenes | Pharmaceuticals |
Each category has distinct chemical structures and market dynamics.
Fluoropolymers
Fluoropolymers are high-performance plastics containing fluorinated carbon backbones.
Common fluoropolymers include:
- PTFE (polytetrafluoroethylene)
- PVDF (polyvinylidene fluoride)
- FEP (fluorinated ethylene propylene)
- PFA (perfluoroalkoxy polymer)
Key Properties of Fluoropolymers
| Property | Performance |
|---|---|
| Temperature resistance | up to 260°C |
| Chemical resistance | Excellent |
| Friction coefficient | Extremely low |
| Electrical insulation | Outstanding |
These characteristics make fluoropolymers essential for aerospace, electronics, and chemical processing equipment.
Fluorinated Refrigerants
Fluorochemicals are widely used as refrigerants due to their stability and thermodynamic efficiency.
Examples include:
| Refrigerant | Chemical Type | Application |
|---|---|---|
| R-134a | HFC | Automotive AC |
| R-1234yf | HFO | Low-GWP refrigerant |
| R-410A | HFC blend | HVAC systems |
Refrigerants represent one of the largest fluorochemical markets.
Fluorinated Pharmaceutical Intermediates
Fluorine is widely used in drug development.
Approximately 20–25% of modern pharmaceuticals contain fluorine atoms.
Examples include:
| Drug Category | Fluorine Function |
|---|---|
| Antiviral drugs | Metabolic stability |
| Antidepressants | Lipophilicity control |
| Cancer treatments | Target specificity |
Fluorine substitution can significantly improve drug performance.
Why Fluorochemicals Have Unique Properties
The extraordinary performance of fluorochemicals comes from molecular physics.
Fluorine’s Unique Characteristics
| Property | Value |
|---|---|
| Atomic number | 9 |
| Electronegativity | 3.98 (highest of all elements) |
| Atomic radius | Very small |
| Reactivity | Extremely high |
When bonded to carbon, fluorine pulls electron density strongly toward itself.
Resulting Molecular Effects
These effects create:
- strong molecular stability
- reduced chemical reactivity
- low intermolecular attraction
Surface Energy Effects
Fluorinated molecules create surfaces that repel both water and oil.
| Material | Surface Energy (mN/m) |
|---|---|
| PTFE | ~18 |
| Polyethylene | ~31 |
| Steel | ~500 |
Low surface energy explains why PTFE coatings are non-stick.
Industrial Applications of Fluorochemicals
Fluorochemicals support a wide range of modern industries.
Electronics and Semiconductors
The semiconductor industry relies heavily on fluorinated gases.
Examples include:
| Gas | Function |
|---|---|
| CF₄ | Plasma etching |
| NF₃ | Chamber cleaning |
| HF | Silicon wafer processing |
Without fluorochemicals, advanced microchip fabrication would be extremely difficult.
Energy Storage and Batteries
Lithium-ion batteries rely on fluorinated materials.
Key fluorochemical components include:
| Component | Fluorochemical |
|---|---|
| Cathode binder | PVDF |
| Electrolyte additives | fluorinated solvents |
| Separator coatings | fluoropolymers |
Electric vehicles are driving significant growth in this sector.
Aerospace and Extreme Environments
Fluoropolymers are essential in aerospace systems.
Applications include:
- fuel system seals
- wire insulation
- high-temperature gaskets
- hydraulic systems
Their resistance to heat and chemicals makes them ideal for aircraft systems.
Production of Fluorochemicals
Manufacturing fluorochemicals requires specialized processes.
Primary Raw Materials
| Raw Material | Role |
|---|---|
| Fluorspar (CaF₂) | Source of fluorine |
| Sulfuric acid | HF production |
| Hydrocarbons | organic backbone |
Fluorspar mining is the starting point of most fluorochemical supply chains.
Production Flow Overview
- Fluorspar → hydrogen fluoride
- HF → fluorinated intermediates
- Intermediates → specialty fluorochemicals
Fluorination Methods
| Method | Description |
|---|---|
| Direct fluorination | elemental fluorine reaction |
| Electrochemical fluorination | electrolysis process |
| Halogen exchange | chlorine replaced by fluorine |
Each method is used for different product types.
Advantages of Fluorochemicals
Fluorochemicals offer several advantages over conventional chemicals.
Performance Advantages
- extreme temperature resistance
- excellent chemical inertness
- low friction
- electrical insulation
- UV resistance
These characteristics explain their widespread use in high-performance applications.
Comparison With Other Chemical Families
| Property | Hydrocarbon | Silicone | Fluorochemical |
|---|---|---|---|
| Heat resistance | Medium | High | Very high |
| Chemical resistance | Low | Medium | Excellent |
| Surface energy | High | Medium | Very low |
Fluorochemicals often outperform other materials in extreme conditions.
Environmental and Regulatory Considerations
Fluorochemicals have also attracted environmental attention.
PFAS Concerns
Certain fluorochemicals fall under the PFAS category.
Key concerns include:
- environmental persistence
- groundwater contamination
- long biological half-life
However, not all fluorochemicals are PFAS.
Regulatory Landscape
| Region | Regulation |
|---|---|
| European Union | PFAS restrictions |
| United States | EPA monitoring |
| Asia | selective regulation |
The regulatory landscape continues to evolve.
Future Trends in Fluorochemicals
The fluorochemicals industry continues to evolve.
Emerging Growth Areas
Key growth sectors include:
- semiconductor manufacturing
- electric vehicles
- hydrogen energy systems
- advanced medical devices
Market Forecast
| Segment | Growth Outlook |
|---|---|
| Fluoropolymers | Strong growth |
| Semiconductor gases | Rapid growth |
| Refrigerants | Transitioning |
| PFAS surfactants | Declining |
These trends indicate that fluorochemicals will remain essential for advanced technology.
Conclusion
Fluorochemicals represent one of the most technologically important classes of chemicals in modern industry. Defined by the presence of carbon-fluorine bonds, these compounds possess extraordinary stability, chemical resistance, and surface properties that make them indispensable across sectors ranging from electronics and pharmaceuticals to aerospace and energy storage. While regulatory scrutiny has increased for certain fluorinated substances, the broader fluorochemicals industry continues to evolve, focusing on high-performance materials and specialized applications where their unique properties cannot easily be replaced.
Work With a Trusted Fluorochemical Supplier
If your project requires high-quality fluorochemicals—whether fluorinated intermediates, specialty fluoropolymers, or industrial fluorinated compounds—choosing the right supplier is critical for both performance and compliance. At Sparrow-Chemical, we supply a wide range of fluorochemicals used in pharmaceuticals, electronics, energy storage, and advanced manufacturing. Our team can help you identify the most suitable fluorochemical solutions for your specific applications. To learn more or request technical support, visit https://sparrow-chemical.com/ and connect with our specialists.







