A lot of people hear “Anhydrous Hydrofluoric Acid” and think of it only as a dangerous acid in a cylinder, but that view is incomplete and commercially misleading. If you do not understand what AHF is actually used for, you can easily misjudge whether you need it, confuse it with ordinary aqueous hydrofluoric acid, choose the wrong grade, or misunderstand why entire industries treat it as a strategic raw material rather than just a hazardous corrosive. That confusion leads to real problems: wrong procurement decisions, poor technical content, mismatched storage systems, bad application positioning, and serious safety errors. The real value of AHF lies in the fact that it is not merely an acid—it is one of the key industrial fluorine feedstocks behind major fluorochemical, refining, metal-processing, and specialty materials value chains.
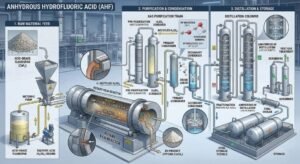
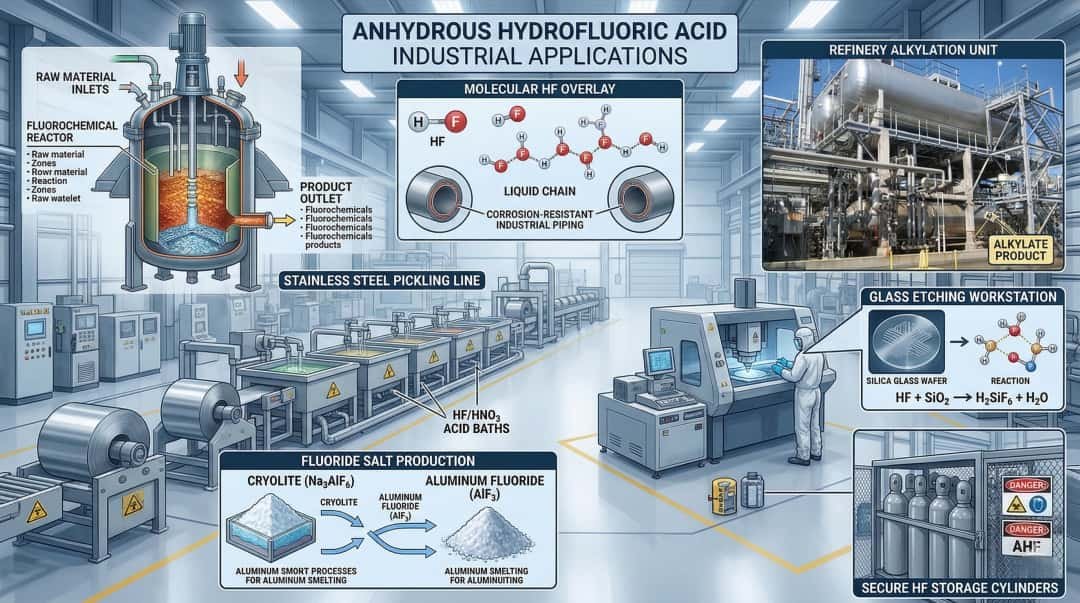
Anhydrous Hydrofluoric Acid (AHF / HF) is used primarily as a high-purity, water-free fluorine feedstock and process acid in major industrial sectors. Its most important uses include making fluorochemicals such as refrigerant intermediates and fluoropolymers, serving as a catalyst in petroleum alkylation for high-octane gasoline components, producing inorganic fluorides used in aluminum and other industries, supporting uranium fluoride production in the nuclear fuel chain, and enabling specialized surface-treatment uses linked to glass etching, metal pickling, oxide removal, and quartz purification. Although hydrofluoric acid solutions are also used in wet cleaning and etching, the anhydrous grade is especially important where low water content, high reactivity, and controlled fluorine chemistry are required.
To understand AHF properly, it helps to stop asking only “What is it?” and start asking “Why do advanced industries still rely on it?” Once you look at its real applications, the picture becomes much clearer: AHF is used where fluorine must be introduced efficiently, where silica-rich materials need chemistry that other acids cannot provide, where catalytic hydrocarbon upgrading is required, and where downstream chemical manufacturing depends on dry hydrogen fluoride rather than diluted hydrofluoric acid. In other words, AHF is best understood as an upstream industrial enabler.
The short answer: what industries use AHF most
Before going deeper, the most practical summary is this: AHF is used where dry hydrogen fluoride chemistry is essential.
The main industrial use groups are:
- Fluorochemical manufacturing
- refrigerant intermediates
- fluorocarbons
- fluoropolymers
- specialty organofluorine compounds
- Petroleum refining
- alkylation catalyst for high-octane gasoline blending components
- Inorganic fluoride production
- aluminum-related fluoride compounds
- fluoride salts
- synthetic cryolite and related materials
- Nuclear fuel processing
- uranium fluoride production
- Specialized materials processing
- glass and silica etching
- quartz purification
- metal pickling and oxide removal
That range already shows why AHF matters commercially. It is not tied to one narrow niche. It serves several industrial systems at once, which is one reason it remains strategically important despite its severe hazard profile. EPA identifies fluorocarbons, aluminum production, uranium processing, petroleum alkylation, and stainless steel pickling among major uses of hydrogen fluoride, while PubChem and Britannica describe HF as a key precursor for numerous commercially important organic and inorganic fluorine compounds.

The most important use of AHF: fluorochemicals
The single most important answer to “What is anhydrous hydrofluoric acid used for?” is fluorochemical production. EPA states that one of the most important uses of hydrogen fluoride is in the production of fluorocarbon chemicals, and notes that a very large share of production is used for this purpose. NCBI’s toxicological production-and-use summary says the most important use of hydrogen fluoride is the production of fluorocarbon chemicals, including hydrofluorocarbons and hydrofluorochlorocarbons. Britannica also states that hydrogen fluoride is employed in the preparation of numerous inorganic and organic fluorine compounds of commercial importance.
Why is AHF used here instead of ordinary aqueous hydrofluoric acid? The answer is simple but important: many fluorochemical pathways need dry HF, not water-rich acid. Water can disrupt reaction selectivity, consume reactive intermediates, increase side reactions, complicate purification, and reduce process efficiency. In fluorochemical plants, AHF is valued because it brings fluorine into the system without bringing in the large water burden associated with aqueous HF. That makes it far more suitable for many industrial fluorination routes, fluoride synthesis steps, and moisture-sensitive downstream chemistries. This is one of the clearest distinctions between anhydrous HF and common hydrofluoric acid solutions used for wet surface treatment.
From a business perspective, this matters even more than many buyers realize. If your company is involved in refrigerant intermediates, fluorinated solvents, fluorinated monomers, fluoropolymer raw materials, or specialty fluorinated building blocks, AHF is often not just an optional reagent; it is part of the foundational upstream chemistry. In that sense, AHF is less like a commodity cleaning acid and more like a platform feedstock. When fluorspar becomes HF, and HF becomes downstream fluorochemicals, an entire value chain is created. That is why AHF often appears close to the beginning of fluorine industry maps.
Key fluorochemical categories linked to AHF
| Downstream category | How AHF is used | Why anhydrous grade matters |
|---|---|---|
| Fluorocarbons | Feedstock for fluorine-containing intermediates and finished compounds | Water control improves process suitability |
| Refrigerant chemistry | Upstream raw material in fluorinated refrigerant chains | Dry HF is important for selective process chemistry |
| Fluoropolymers | Helps enable fluoride-containing monomers and intermediates | Purity and low moisture support downstream quality |
| Specialty organofluorines | Building-block chemistry for high-value compounds | Better suited to moisture-sensitive synthesis |
| Inorganic fluorides | Direct fluoride source for salts and industrial compounds | High active HF content and process consistency |
Source basis: EPA, PubChem, Britannica, NCBI.
AHF in petroleum refining: alkylation service
One of the most commercially important non-fluorochemical uses of AHF is petroleum alkylation. EPA explicitly lists petroleum alkylation among major hydrogen fluoride uses, and AEGL documentation from EPA also describes HF as a catalyst in alkylation processes during petroleum refining. In this application, AHF is not mainly being consumed as a bulk reagent in the same way as a simple raw material; it is being used as a catalyst to help convert light hydrocarbons into high-octane alkylate for gasoline blending. (US EPA)
This is one of the best examples of why the phrase “used for” can mean very different things with AHF. In fluorochemical production, AHF is often used as a fluorine feedstock. In alkylation, it is used because it creates the reaction environment that makes the process economically valuable. The practical output is high-octane gasoline blendstock, not a fluorinated product. That distinction matters for technical writing and customer communication, because the same AHF can serve as a reagent in one industry and as a catalyst in another. (US EPA)
For refineries, AHF’s value in alkylation comes from its strong catalytic performance and process maturity. That does not remove the very serious safety concerns surrounding HF alkylation units, but it explains why the technology persisted for decades in parts of the refining industry. In commercial terms, this means AHF demand is not driven only by specialty chemistry or fluorochemicals; it is also linked to fuel production and refining infrastructure. That broadens its industrial relevance considerably. (US EPA)
What AHF does in alkylation, in simple terms
- It helps create the reaction conditions needed for alkylation
- It supports formation of high-octane alkylate
- It upgrades light hydrocarbons into more valuable gasoline components
- It is used as a catalyst rather than merely as a corrosive process liquid
That catalytic role is a big reason why AHF should never be described narrowly as “just an acid.” In refining, it is part of a major conversion technology. (US EPA)
AHF in aluminum and inorganic fluoride chemistry
Another major industrial use of AHF is the production of inorganic fluorides, especially materials connected to aluminum processing and other fluoride-based industries. EPA’s AP-42 document identifies aluminum production as the largest use for HF in one major usage breakdown, and Britannica specifically gives sodium aluminum fluoride as an example of a commercially important compound prepared from hydrogen fluoride. EPA’s AEGL document also mentions the manufacture of artificial cryolite and fluoride salts among HF uses. (US EPA)
This application group is easy to underestimate because it sounds less visible than refrigerants or gasoline. In reality, it is one of the clearest demonstrations of AHF’s role as a core fluorine building block. Here AHF is used to make fluoride-bearing inorganic materials that serve metallurgical and industrial purposes. That includes aluminum-related fluoride chemistry, synthetic cryolite, and other fluoride salts used across different processes. The exact downstream product varies, but the common logic is the same: AHF provides reactive fluorine in a form suitable for inorganic chemical conversion at industrial scale. (Encyclopedia Britannica)
For buyers and technical marketers, this has an important implication. When your customer asks about AHF, they may not be asking about etching or cleaning at all. They may be part of an upstream fluoride value chain, a metallurgical chemicals chain, or a producer of process salts and industrial fluorides. If your product page only talks about hazard and glass etching, you miss much of the real market. AHF is used far more broadly than that. (US EPA)
AHF in uranium processing and the nuclear fuel chain
One of the most strategic uses of AHF is in uranium chemistry, particularly in the conversion of uranium compounds into uranium fluorides. EPA lists uranium processing among major hydrogen fluoride uses, and the AEGL document notes production of uranium hexafluoride as an HF-related application. This makes AHF relevant not just to chemicals and refining, but also to a tightly controlled and geopolitically sensitive segment of the nuclear fuel cycle. (US EPA)
This application is important for two reasons. First, it demonstrates that AHF is not merely a bulk process chemical for general industry; it is also part of high-consequence strategic supply chains. Second, it shows why purity, dryness, contamination control, and secure logistics can be so important in AHF trade. In nuclear-related chemical pathways, product quality and handling discipline matter even more than in many ordinary commodity chemical transactions. AHF is used here because fluoride chemistry is necessary to move uranium into chemically useful forms for subsequent fuel-cycle operations. (US EPA)
For content strategy, this means that AHF belongs in discussions of critical industrial materials, not just hazardous acids. Its application footprint reaches into sectors that are capital-intensive, regulated, and strategically significant. That alone should tell buyers and marketers that this material occupies a different category from ordinary mineral-acid products. (US EPA)
One of the most famous uses of HF chemistry is glass etching, and this remains a core part of how the public and many industrial users understand the material. EPA states that aqueous hydrofluoric acid is used in glass etching and quartz purification, while PubChem also notes hydrofluoric acid is used mainly for etching glass. Britannica’s historical discussion of HF corroding glass apparatus reflects the same underlying chemistry: HF is unusual among acids because it reacts effectively with silica-containing materials. (US EPA)
Here it is important to draw a careful distinction. The broader HF family of uses absolutely includes glass etching and silica removal. However, not every glass-etching operation uses fully anhydrous HF; many rely on aqueous HF or formulated etchants. Still, AHF is commercially relevant to this sector because it is the upstream dry hydrogen fluoride source from which many downstream HF products and etching chemistries ultimately derive. So when asking what AHF is used for, the technically correct answer includes both direct and value-chain-linked roles: AHF is used directly in dry industrial chemistry and indirectly as the base feedstock supporting many HF-derived surface-treatment applications. (US EPA)
In practical industrial terms, silica-related HF uses include:
- decorative and technical glass etching
- quartz purification
- oxide and silicate removal
- certain electronics and materials-cleaning applications
- removal of silica-rich residues that many other acids cannot dissolve effectively
That last point is especially important. AHF and HF chemistry are not valued here simply because they are corrosive. They are valued because they solve a chemistry problem that many common acids do not solve well: silica attack. That is why HF remains distinctive in materials processing. (US EPA)
AHF in stainless steel pickling, oxide removal, and metal treatment
Another practical use area is metal treatment, especially stainless steel pickling and oxide removal. EPA names stainless steel pickling among the major uses of HF. In these applications, HF chemistry helps remove oxide scales and difficult surface films, often in combination with other acids depending on the process. (US EPA)
This is another place where non-specialists often misunderstand the role of AHF. The value is not that AHF is a generic “stronger acid cleaner.” The value is that HF chemistry can attack oxide and siliceous surface components that are resistant to many other systems. In stainless steel finishing and related metal-processing applications, this makes HF-containing chemistries especially useful for descaling, cleaning, and preparing the surface for further treatment or service. (US EPA)
Again, not every metal-treatment application uses pure anhydrous HF directly. Many use aqueous hydrofluoric acid or mixed-acid systems. But AHF still matters to this application family because it is the upstream hydrogen fluoride product and, in some specialized process environments, the low-water form is preferred or necessary. For industrial content, the correct phrasing is that AHF is used directly in dry fluoride chemistry and indirectly underpins multiple HF-based metal-treatment uses. (US EPA)
AHF as the upstream platform for multiple downstream sectors
One of the best ways to answer the user’s question is not by listing isolated uses, but by showing the application structure more clearly. AHF is best understood as an upstream platform chemical that feeds multiple downstream sectors.
Application structure map
| Application tier | Main use of AHF | Typical downstream outcome |
|---|---|---|
| Upstream fluorine feedstock | Organic and inorganic fluorine chemistry | Fluorochemicals, fluorides, specialty intermediates |
| Catalyst use | Petroleum alkylation | High-octane alkylate for gasoline blending |
| Inorganic conversion | Fluoride salts, cryolite, aluminum-related compounds | Metallurgical and industrial process materials |
| Nuclear chemistry | Uranium fluoride production | Nuclear fuel-cycle intermediates |
| Surface chemistry support | Glass etching, oxide removal, quartz purification | Materials processing and surface treatment |
Source basis: EPA, PubChem, Britannica, NCBI. (US EPA)
Seen this way, AHF has five major commercial identities at once:
- a feedstock
- a catalyst
- an inorganic fluoride precursor
- a strategic nuclear-processing reagent
- an enabler of silica and oxide treatment chemistry
That is a much better industrial description than simply calling it “a dangerous acid.” (US EPA)
Why industries prefer anhydrous HF instead of aqueous HF in some applications
This is the part many product pages fail to explain clearly. The reason some industries specifically use anhydrous HF rather than ordinary hydrofluoric acid solution is that the absence of water is itself a technical advantage.
The main reasons are:
- Moisture-sensitive reaction pathways
- water can reduce yield or generate side reactions
- Better suitability for fluorochemical synthesis
- dry HF is often the preferred fluorine input
- Catalytic requirements
- alkylation service uses anhydrous HF, not a dilute cleaning acid
- Controlled impurity profile
- process quality may depend on dryness and purity
- Different handling and reaction behavior
- anhydrous HF behaves differently from aqueous HF in both chemistry and equipment design
Britannica explains that hydrogen fluoride is obtained in a fairly anhydrous state by fractional distillation and stored in steel cylinders, which reflects how industry intentionally produces and preserves this low-water form for commercial use. (Encyclopedia Britannica)
This also explains a frequent buyer mistake: assuming that “HF is HF.” It is not. In many industrial applications, 49% HF and anhydrous HF are not commercially interchangeable, even though they are related substances. The intended use determines which form is appropriate. AHF is used where dry-process chemistry matters; aqueous HF is more common where wet etching or wet pickling chemistry is acceptable or preferred. (US EPA)
The difference between direct uses and indirect value-chain uses
To write accurately about AHF, it helps to separate two categories:
Direct uses of AHF
These are cases where the anhydrous material itself is intentionally used in the process.
Examples include:
- fluorochemical feedstock applications
- petroleum alkylation
- inorganic fluoride production
- uranium fluoride production
- selected moisture-sensitive specialty syntheses
Indirect value-chain uses of AHF
These are sectors where AHF is the upstream source that supports later HF-derived or fluoride-derived applications.
Examples include:
- some glass etching product systems
- quartz purification chemicals
- metal pickling formulations
- certain cleaning and oxide-removal chemistries
- some electronics and materials-processing HF products
This distinction matters because it avoids two common errors. The first error is overstating that every HF-related application uses pure AHF directly. The second error is understating AHF’s importance by only counting direct uses. AHF’s real industrial footprint includes both. (US EPA)
Where AHF creates the most industrial value
If the question is not only “What is AHF used for?” but “Where does it create the most industrial value?” the answer is more strategic.
AHF creates the highest industrial value where it enables:
- high-value fluorochemical production
- essential refinery upgrading
- metallurgical fluoride chemistry
- critical nuclear conversion chemistry
- surface-treatment functions that competing acids cannot match
That value comes from chemical uniqueness. HF is one of the most important bridges between mineral fluorine and commercially useful fluorinated products. It is also one of the few widely established chemistries that can economically serve such different sectors at once. That is why AHF remains important even though it is difficult and dangerous to handle. (Encyclopedia Britannica)
Industries that commonly care about AHF specifications
Different application sectors care about different things. A more useful buying-oriented view looks like this:
| Industry / buyer type | What they mainly use AHF for | What they care about most |
|---|---|---|
| Fluorochemical producers | Feedstock for fluorinated compounds | Purity, water content, stable supply |
| Refiners | Alkylation catalyst | reliability, containment discipline, process compatibility |
| Inorganic fluoride producers | Fluoride salts and metallurgical compounds | active HF content, lot consistency |
| Nuclear-related processors | Uranium fluoride chemistry | purity, trace impurities, secure logistics |
| Metal-treatment producers | HF-based process chemistry | concentration fit, application match |
| Glass / quartz processors | Etching and purification chemistry | reaction performance, formulation suitability |
This is a more commercial way to describe use cases, and for many B2B customers it is more helpful than long theoretical chemistry paragraphs. The “used for” question is usually really asking: used by whom, for what result, and under what technical requirements? (US EPA)
What AHF is not mainly used for
This is just as important as listing its uses. AHF is not mainly used as:
- a routine household acid
- a general-purpose industrial cleaner like hydrochloric acid
- a casual laboratory acid for normal use
- a commodity wet acid for every surface-treatment job
- an interchangeable substitute for all HF solution grades
The reason this matters is simple: when companies describe AHF too loosely, they attract the wrong traffic, confuse buyers, and create technical misunderstandings. The real market for AHF is specialized, high-consequence, and application-specific. Its uses are industrially significant, but they are not generic. (US EPA)
Why understanding AHF uses also means understanding AHF hazards
Any honest article about AHF applications has to acknowledge that its uses cannot be separated from its hazard profile. CDC states that hydrofluoric acid is corrosive and causes destruction of deep tissues when fluoride ions penetrate the skin, and substantial absorption may be fatal. ATSDR notes rapid penetration of fluoride into tissue and reports systemic poisoning after dermal exposure to anhydrous HF. The American Chemistry Council’s emergency first-aid guidance for AHF and HF makes the same point: HF differs from many acids because fluoride penetrates tissue rapidly and can continue damaging deep layers. (wwwn.cdc.gov)
Why is that relevant in an “applications” article? Because some of AHF’s uses exist precisely because the chemical is unusually reactive and chemically distinctive. The same properties that make it valuable in fluorochemical feedstock chemistry, silica attack, and catalytic service also make it exceptionally unforgiving in exposure scenarios. So the best practical answer is this: AHF is used where its unique chemistry justifies the burden of specialized handling. Industries do not choose it casually. They choose it when other chemicals cannot deliver the same result, or cannot deliver it economically at scale. (US EPA)
A practical decision framework: do you really need AHF?
For readers thinking commercially, this may be the most useful part of the article. When evaluating AHF, ask the following questions:
You likely need AHF if:
- your process requires dry HF
- water contamination harms yield or selectivity
- you are producing fluorochemicals or inorganic fluorides
- you are working in HF alkylation or similar catalytic systems
- you need an upstream fluorine raw material rather than a wet etching acid
You may need aqueous HF instead if:
- your use is mainly wet etching
- your use is stainless steel pickling or surface treatment
- your process is solution-based and water is acceptable
- you are not doing moisture-sensitive fluorine chemistry
You should rethink the project if:
- the site lacks HF-specific handling capability
- the process only needs a generic acid
- the buyer is treating AHF as a routine bulk corrosive
- the team cannot clearly define purity, water, and application requirements
That kind of structured evaluation is far more useful to industrial readers than simply repeating that AHF is “used in many industries.” (US EPA)
Final answer
Anhydrous Hydrofluoric Acid is used mainly as a dry hydrogen fluoride feedstock and process chemical in advanced industry. Its biggest uses are producing fluorochemicals, serving as a catalyst in petroleum alkylation, making inorganic fluoride compounds for aluminum and related sectors, supporting uranium fluoride production, and enabling HF-based glass, quartz, oxide, and metal-treatment chemistries. What makes AHF special is not that it is simply a powerful acid, but that it provides industrial fluorine in a low-water form that many important chemical and catalytic processes require. (US EPA)
So if someone asks, “What is AHF used for?” the most accurate answer is this: it is used where industries need reactive fluorine, dry-process HF chemistry, or catalytic and materials-processing performance that ordinary acids cannot provide. That is why AHF remains one of the most important upstream materials in the fluorine industry, even though it is also one of the most hazardous acids in routine industrial service. (wwwn.cdc.gov)
A practical note before you source or position AHF
If you are building an AHF product page, qualifying suppliers, or evaluating whether your application truly needs Anhydrous Hydrofluoric Acid instead of aqueous hydrofluoric acid, the key is to define the real use case first: fluorochemical feedstock, alkylation catalyst, inorganic fluoride production, nuclear chemistry, or HF-based surface-treatment value chain. At Sparrow-Chemical, we focus on application-fit chemical communication and practical industrial sourcing logic. For fluorochemical raw materials, HF-related product positioning, or specification discussions, visit Sparrow-Chemical: https://sparrow-chemical.com/